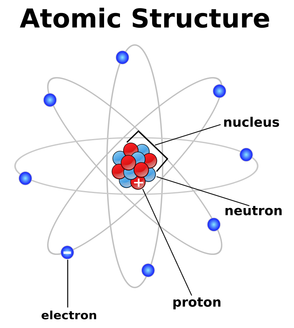
An atom is the basic structure/building block of all matter. An atom is made up of protons(+), electrons(-), and neutrons(ø). The number of protons in the nucleus equals the atom's atomic number, and the number of neutrons and protons added together equals the atom's atomic mass. Electrons surround the nucleus and there are normally the same amount of electrons as protons. Protons and neutrons have mass and electrons do not. An isotope is an atom with an unusual number of neutrons, and an ion is an atom with an unusual number of electrons. A molecule is two or more atoms boded together(usually through shared electrons). Some examples of molecules are H20, CO2, etc. A compound is a molecule made up of different elements. An example of a compound is CO2.
Solids are molecules that are bonded firmly to one another and held densely. Liquids are molecules that are bonded to one another, but can change which molecules they are bonded to.Gases are molecules that are very rarely bonded to one another. Gases have the most energy and heat compared to a solid, which has the least amount of energy and heat.
The colder it is, the slower molecules move. Their volume decreases along with their elevation. The pressure increases and so does the density. The hotter it is, the faster molecules move. The volume and elevation increases, and the pressure and density decreases.
Solids are molecules that are bonded firmly to one another and held densely. Liquids are molecules that are bonded to one another, but can change which molecules they are bonded to.Gases are molecules that are very rarely bonded to one another. Gases have the most energy and heat compared to a solid, which has the least amount of energy and heat.
The colder it is, the slower molecules move. Their volume decreases along with their elevation. The pressure increases and so does the density. The hotter it is, the faster molecules move. The volume and elevation increases, and the pressure and density decreases.