The goal for this project was to create a board game for ages 10+ that consists of chemical and physical changes. It needs to be entertaining for players, but not too challenging that it becomes frustrating for them. Blueprints of the board were required along with detailed instructions on how to play and set up the game. Detailed safety precautions should also be provided. It also needs to have a clear, consistent theme that is fun for all who play.
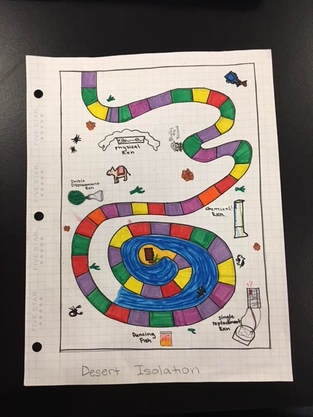
My group and I decided on a desert treasure theme for our board game. In this game, you go on an exciting desert journey filled with many thrilling obstacles. The goal of the game is to be the first player to reach the hidden treasure chest in the desert.
My group and I decided on a desert treasure theme for our board game. In this game, you go on an exciting desert journey filled with many thrilling obstacles. The goal of the game is to be the first player to reach the hidden treasure chest in the desert.
The Game
Safety Precautions
Board Game Blueprint
The Chemistry
Double Displacement Reaction: sodium bicarbonate (baking soda) and dilute acetic acid (vinegar)
NaHCO3(s) + CH3COOH(l) → CO2(g) + H2O(l) + Na+(aq) + CH3COO-(aq)
A double displacement reaction is where two aqueous ionic compounds exchange ions and form two new ionic compounds. It goes from two aqueous and aqueous to aqueous and either a solid, liquid, or gas. When this happens, it forms a precipitate. Acetic acid and sodium bicarbonate react in a double displacement reaction to form sodium acetate and carbonic acid. The carbonic acid then has a decomposition reaction and produces carbon dioxide gas which causes the solutions to bubble. Water and oxygen are also produced from this reaction. The release of the carbon dioxide gas flows out of the flask and into the balloon, inflating it.
Single Replacement Reaction: Aluminum and Copper (II) chloride
3CuCl2(aq) + 2Al(s) → 2AlCl3 + 3Cu(s)
A single replacement reaction is where an element replaces its like element in an ionic compound or acid. Metals always replace the cations and the nonmetals always replace the anions. The reaction will only occur if the element is more reactive than its like element in the compound. The aluminum and copper react to create a single replacement reaction. The aluminum, a metal, replaces the cation, copper (II), causing the aluminum foil to break down, as it forms aluminum chloride. Aluminum has a higher oxidation base, meaning it is more reactive, than the aqueous copper (II) chloride solution. This results in a reaction that breaks down the aluminum foil. When the foil is taken out of the circuit, the electricity takes the path through the light to light up the flare.
Chemical Change (Decomposition): hydrogen peroxide, potassium iodide, and dish soap
2H2O2(aq) → 2H2O(l) + O2(g)
The hydrogen peroxide is a decomposition reaction, where one reactant yields two or more products. The potassium iodide reacts with the hydrogen peroxide by breaking it down into oxygen and water. The dish soap captures the oxygen gas in bubbles, which also means a flame will grow when placed in the foam because of the oxygen gas stored in the bubbles. The foam is formed from the soap. Iodine is also produced, which is why the foam has a yellow tinge. This is an exothermic reaction where heat is released, so you may see steam and feel heat -- be careful!
Physical Change (Solid to Gas): dry ice, soap, and water
H2O + CO2 → H2CO3
The dry ice consists of frozen CO2. The water reacts with the ice, causing it to turn quickly into a gaseous form. This quick phase change causes a lot of steam to be released, creating the dense fog effect.
Alka-Seltzer Tablets, Water, and Oil
The alka-seltzer tablets are chemicals in solid form. When placed in the water, the alka-seltzer tablet dissolves due to the chemicals being released. It undergoes a reaction and changes from a solid to a liquid. Bubbles are produced. The oil swirls around and does not mix into the water because it is nonpolar. Since water is hydrophilic and oil is hydrophobic, they do not combine.
NaHCO3(s) + CH3COOH(l) → CO2(g) + H2O(l) + Na+(aq) + CH3COO-(aq)
A double displacement reaction is where two aqueous ionic compounds exchange ions and form two new ionic compounds. It goes from two aqueous and aqueous to aqueous and either a solid, liquid, or gas. When this happens, it forms a precipitate. Acetic acid and sodium bicarbonate react in a double displacement reaction to form sodium acetate and carbonic acid. The carbonic acid then has a decomposition reaction and produces carbon dioxide gas which causes the solutions to bubble. Water and oxygen are also produced from this reaction. The release of the carbon dioxide gas flows out of the flask and into the balloon, inflating it.
Single Replacement Reaction: Aluminum and Copper (II) chloride
3CuCl2(aq) + 2Al(s) → 2AlCl3 + 3Cu(s)
A single replacement reaction is where an element replaces its like element in an ionic compound or acid. Metals always replace the cations and the nonmetals always replace the anions. The reaction will only occur if the element is more reactive than its like element in the compound. The aluminum and copper react to create a single replacement reaction. The aluminum, a metal, replaces the cation, copper (II), causing the aluminum foil to break down, as it forms aluminum chloride. Aluminum has a higher oxidation base, meaning it is more reactive, than the aqueous copper (II) chloride solution. This results in a reaction that breaks down the aluminum foil. When the foil is taken out of the circuit, the electricity takes the path through the light to light up the flare.
Chemical Change (Decomposition): hydrogen peroxide, potassium iodide, and dish soap
2H2O2(aq) → 2H2O(l) + O2(g)
The hydrogen peroxide is a decomposition reaction, where one reactant yields two or more products. The potassium iodide reacts with the hydrogen peroxide by breaking it down into oxygen and water. The dish soap captures the oxygen gas in bubbles, which also means a flame will grow when placed in the foam because of the oxygen gas stored in the bubbles. The foam is formed from the soap. Iodine is also produced, which is why the foam has a yellow tinge. This is an exothermic reaction where heat is released, so you may see steam and feel heat -- be careful!
Physical Change (Solid to Gas): dry ice, soap, and water
H2O + CO2 → H2CO3
The dry ice consists of frozen CO2. The water reacts with the ice, causing it to turn quickly into a gaseous form. This quick phase change causes a lot of steam to be released, creating the dense fog effect.
Alka-Seltzer Tablets, Water, and Oil
The alka-seltzer tablets are chemicals in solid form. When placed in the water, the alka-seltzer tablet dissolves due to the chemicals being released. It undergoes a reaction and changes from a solid to a liquid. Bubbles are produced. The oil swirls around and does not mix into the water because it is nonpolar. Since water is hydrophilic and oil is hydrophobic, they do not combine.
Chemical Change Physical Change Double Displacement Reaction Alka-Seltzer Tablets
|
|
|
|
|
Concepts
Element: A substance that cannot be broken down into simpler substances.
Atoms: The smallest unit of an element that maintains its chemical properties; made up of protons, neutrons, and electrons.
Electrons (e⁻): They make up atoms and are located outside the nucleus. They have a negative charge and make up the atom's volume.
Neutrons (n⁰): They make up atoms and are located in the nucleus. They have a neutral charge and make up the atom's mass.
Protons (p⁺): They make up atoms and are located in the nucleus. They have a positive charge, make up the atom's mass, is the atomic number, and determines the type of element.
Ions: An atom that has gained or lost electrons, giving it a positive or negative charge.
Polyatomic Ions:
Cations: An ion with a positive charge.
Anions: An ion with a negative charge; nonmetals.
Chemical Reactions: A process where one or more substances change change to produce different substances.
Physical Change: A change in the physical properties of the substance. The substance does not chemically change.
Synthesis Reaction: Two or more reactants combine to form one product. A+B-->AB
Decomposition Reaction: One reactant yields two or more products AB-->A+B
Single Replacement Reaction: An element replaces its like element in an ionic compound or element. The reaction will go if the element is more reactive than its like element in the compound. AB(aq) + C --> CB(aq) +A *Metals replace cations and nonmetals replace anions
Double Displacement Reaction: Two aqueous ionic compounds exchange ions and form two new ionic compounds. Will only go if one of the products is either a solid (precipitate), water, or gas. AB(aq) + CD(aq) --> AD(aq) + CB(s)
Combustion Reaction: Carbon dioxide and water are always products. Hydrocarbon + 02(g) --> CO2(g) + H2O(g)
Exothermic Reaction: A reaction where heat is released.
Solid (s): A state of matter where the atoms are tightly bonded, giving it a definite shape and volume.
Gas (g): A state of matter where the atoms are more spaced out, giving it a definite volume but not shape.
Liquid (l): A state of matter where the atoms are spaced out, giving it neither a definite shape or volume.
Aqueous (aq): A solvent is dissolved in water.
Precipitate: A substance that forms a solid from a solution due to a double displacement reaction.
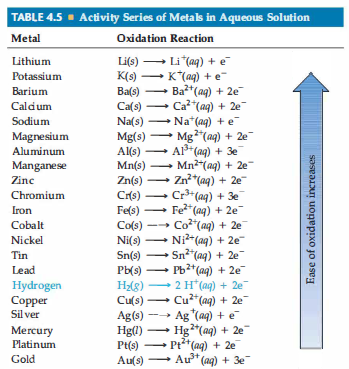
Activity Series: A series of elements with similar properties that are arranged in descending order of chemical activity; shows how reactive they are.
Solubility Guidelines: Guidelines that help determine what substances are soluble.
Soluble: A substance that can dissolve in water.
Chemical Equations: A representation of a chemical reaction that uses symbols to show relationships. The equation must be balanced.
Atoms: The smallest unit of an element that maintains its chemical properties; made up of protons, neutrons, and electrons.
Electrons (e⁻): They make up atoms and are located outside the nucleus. They have a negative charge and make up the atom's volume.
Neutrons (n⁰): They make up atoms and are located in the nucleus. They have a neutral charge and make up the atom's mass.
Protons (p⁺): They make up atoms and are located in the nucleus. They have a positive charge, make up the atom's mass, is the atomic number, and determines the type of element.
Ions: An atom that has gained or lost electrons, giving it a positive or negative charge.
Polyatomic Ions:
Cations: An ion with a positive charge.
Anions: An ion with a negative charge; nonmetals.
Chemical Reactions: A process where one or more substances change change to produce different substances.
Physical Change: A change in the physical properties of the substance. The substance does not chemically change.
Synthesis Reaction: Two or more reactants combine to form one product. A+B-->AB
Decomposition Reaction: One reactant yields two or more products AB-->A+B
Single Replacement Reaction: An element replaces its like element in an ionic compound or element. The reaction will go if the element is more reactive than its like element in the compound. AB(aq) + C --> CB(aq) +A *Metals replace cations and nonmetals replace anions
Double Displacement Reaction: Two aqueous ionic compounds exchange ions and form two new ionic compounds. Will only go if one of the products is either a solid (precipitate), water, or gas. AB(aq) + CD(aq) --> AD(aq) + CB(s)
Combustion Reaction: Carbon dioxide and water are always products. Hydrocarbon + 02(g) --> CO2(g) + H2O(g)
Exothermic Reaction: A reaction where heat is released.
Solid (s): A state of matter where the atoms are tightly bonded, giving it a definite shape and volume.
Gas (g): A state of matter where the atoms are more spaced out, giving it a definite volume but not shape.
Liquid (l): A state of matter where the atoms are spaced out, giving it neither a definite shape or volume.
Aqueous (aq): A solvent is dissolved in water.
Precipitate: A substance that forms a solid from a solution due to a double displacement reaction.
Activity Series: A series of elements with similar properties that are arranged in descending order of chemical activity; shows how reactive they are.
Solubility Guidelines: Guidelines that help determine what substances are soluble.
Soluble: A substance that can dissolve in water.
Chemical Equations: A representation of a chemical reaction that uses symbols to show relationships. The equation must be balanced.
Reflection
This was a very fun project. One thing my group and I did an excellent job of was collaborating. We were all comfortable sharing ideas and building off of each others thoughts. We were supportive of each other and open to ideas. Because of this, we were able to make decisions quickly and decide on things together without having any conflicts. Another thing I did well on for this project was being optimistic. I made sure to stay positive and supportive even when my group was having a difficult time figuring things out. One issue my group and I had was with our time management. We spent more time painting the board and adding on fun details, which gave us less time to plan out and test our chemical reactions. One thing I could work on for next time is being more of a leader. This way I can make sure all of the group members have something to work on so we won't be rushed to finish the project on time. A second thing I need to work on are my presentation skills. Though my group and I didn't present in front of the class, I still felt very reliant on my notes while filing our presentation. I can fix this by making sure I fully understand the concepts I am presenting and practice a few times beforehand. This will make me more confident and knowledgable when I present. Overall, I thought this was a successful project. My group and I ended up constructing a fun and unique board game that was entertaining to make and play.